 /studio-shot-of-chest-x-ray-136595862-595bdb493df78c4eb6a38dce-5c59d3ecc9e77c000132accf.jpg)
Also, in contrast to nuclear scattering, the magnetic form factor is not isotope dependent, but is dependent on the oxidation state of the atom. Hence, in strong contrast to the case for nuclear scattering, the scattering object for magnetic scattering is far from a point source it is still more diffuse than the effective size of the source for X-ray scattering, and the resulting Fourier transform (the magnetic form factor) decays more rapidly than the X-ray form factor. However, this neutron-magnetic scattering is only from the outer electrons, rather than being heavily weighted by the core electrons, which is the case for X-ray scattering. In an X-ray diffraction experiment a set of crystal lattice planes (hkl) is selected by the incident conditions and the lattice spacing d hkl is determined through the well-known Braggs law. 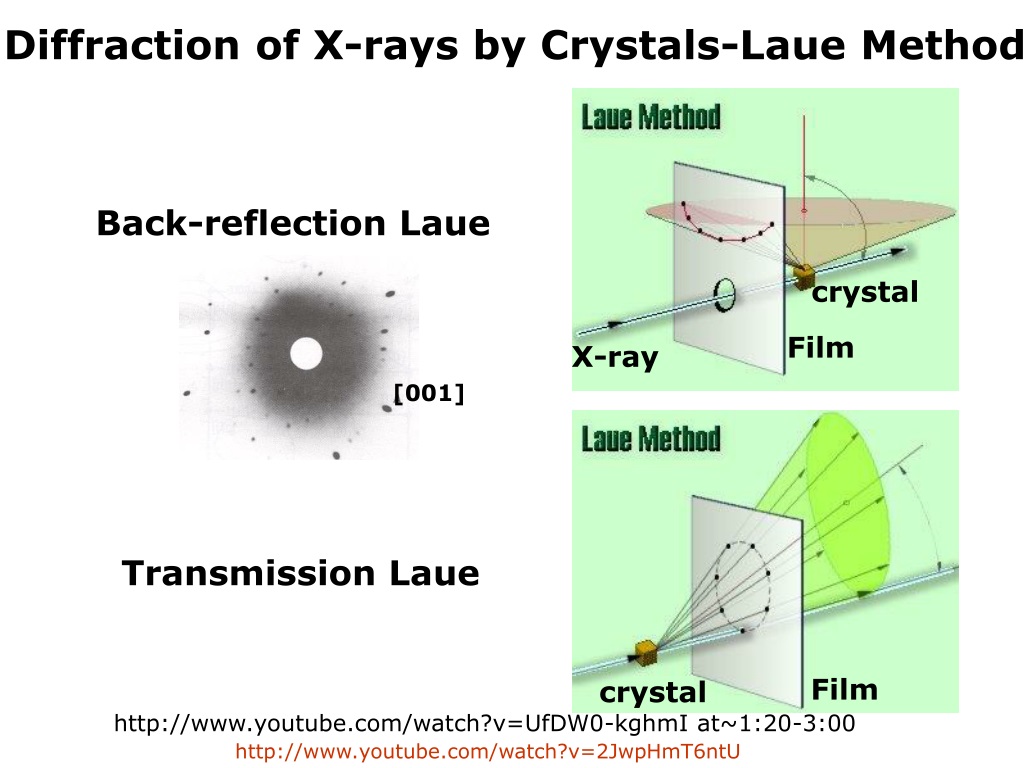
Since these orbitals are typically of a comparable size to the wavelength of the free neutrons, the resulting form factor resembles that of the X-ray form factor. 
For an object with spatial density distribution, ρ ( r ) for magnetic scattering. The common feature of all form factors is that they involve a Fourier transform of a spatial density distribution of the scattering object from real space to momentum space (also known as reciprocal space). The atomic form factor depends on the type of scattering, which in turn depends on the nature of the incident radiation, typically X-ray, electron or neutron. Consequently, the X-ray diffraction pattern is the fingerprint of periodic atomic. The peak intensities are determined by the distribution of atoms within the lattice. In physics, the atomic form factor, or atomic scattering factor, is a measure of the scattering amplitude of a wave by an isolated atom. X-ray diffraction peaks are produced by constructive interference of a monochromatic beam of X-rays scattered at specific angles from each set of lattice planes in a sample. X-ray atomic form factors of oxygen (blue), chlorine (green), Cl − (magenta), and K + (red) smaller charge distributions have a wider form factor. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
